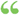 As used in this subsection . . ., the term 'substantial evidence' means evidence consisting of adequate and well-controlled investigations, including clinical investigations, by experts qualified by scientific training and experience to evaluate the effectiveness... As used in this subsection . . ., the term 'substantial evidence' means evidence consisting of adequate and well-controlled investigations, including clinical investigations, by experts qualified by scientific training and experience to evaluate the effectiveness...  FDA Proposals to Permit the Use of Disease-specific Health Claims on Food ... - Page 348de United States. Congress. House. Committee on Government Operations. Human Resources and Intergovernmental Relations Subcommittee - 1988 - 471 pagesAffichage du livre entier FDA Proposals to Permit the Use of Disease-specific Health Claims on Food ... - Page 348de United States. Congress. House. Committee on Government Operations. Human Resources and Intergovernmental Relations Subcommittee - 1988 - 471 pagesAffichage du livre entier - À propos de ce livre
 | United States - 1964 - 1098 pages
...As used in this subsection and subsection (e) of this section, the term "substantial evidence" means evidence consisting of adequate and wellcontrolled...drug involved, on the basis of which it could fairly TITLE 21.— POOD AND DRUGS Page 1142 and responsibly be concluded by such experts that the drug will... | |
 | United States - 1977 - 1276 pages
...adequate and well-controlled investigations, including field investigation, by experts qualified by scientific training and experience to evaluate the effectiveness of the drug involved, on thé basis of which it could fairly and reasonably be concluded by such experts that the drug will... | |
 | 1964 - 1242 pages
...to the Food and Drug Administration. b. An application may be refused unless It Includes substantial evidence consisting of adequate and well-controlled...the conditions of use prescribed, recommended, or suggested In the proposed labeling. c. The complete composition and/or method of manufacture of the... | |
 | 1990 - 1086 pages
...adequately ensure safety and efficacy of use. "Substantial evidence" has been defined by Congress to mean "evidence consisting of adequate and well-controlled...purports or is represented to have under the conditions prescribed, recommended, or suggested in the labeling or proposed labeling thereof." This definition... | |
 | 1998 - 1162 pages
...adequately ensure safety and efficacy of use. "Substantial evidence" has been defined by Congress to mean "evidence consisting of adequate and well-controlled...purports or is represented to have under the conditions prescribed, recommended, or suggested in the labeling or proposed labeling thereof." This definition... | |
 | 1998 - 1120 pages
...adequately ensure safety and efficacy of use. "Substantial evi-dence" has been defined by Congress to mean "evidence consisting of adequate and well-controlled...purports or is represented to have under the conditions prescribed, rec-ommended, or suggested in the labeling or proposed labeling thereof." This def-inition... | |
 | 1986 - 1610 pages
...adequately ensure safety and efficacy of use. "Substantial evidence" has been defined by Congress to mean "evidence consisting of adequate and well-controlled...purports or is represented to have under the conditions prescribed, recommended, or suggested in the labeling or proposed labeling thereof." This definition... | |
 | 1977 - 1080 pages
...may be refused unless It Includes substantial evidence consisting of adequate and well -con trolled Investigations, Including clinical Investigations,...the conditions of use prescribed, recommended, or suggested In the proposed labeling. c. Reports of all clinical tests sponsored by the applicant or... | |
 | 1981 - 1252 pages
...adequately insure safety and efficacy of use. "Substantial evidence" has been defined by Congress to mean "evidence consisting of adequate and well-controlled...concluded by such experts that the drug will have the effectiveness it purports and is represented to have under the conditions prescribed, recommended or... | |
 | 1978 - 860 pages
...adequately Insure safety and efficacy of use. Substantial evidence” has been defined by Congress to mean “evidence consisting of adequate and well-controlled...responsibly be concluded by such experts that the drug vill have the effectiveness It purports and is represented to have under the conditions prescribed,... | |
| |